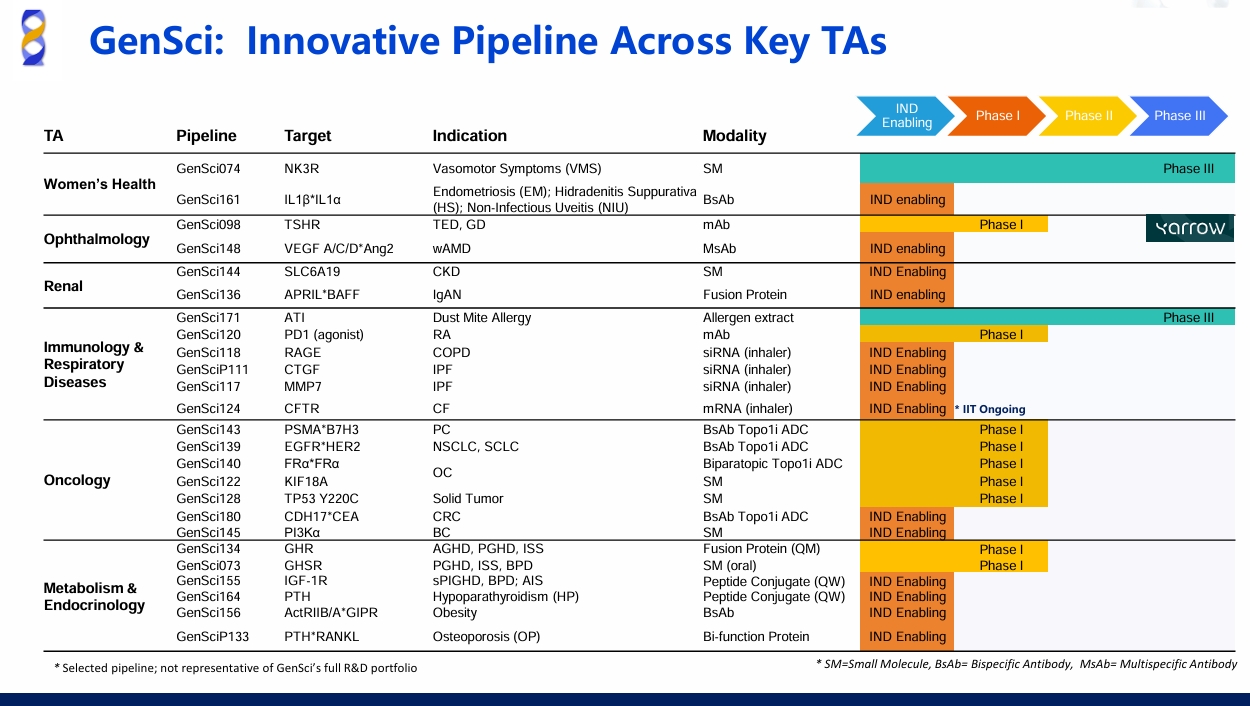
GenSci’s NK3R Antagonist for VMS Phase II Study Results Published in Menopause
Time
2026-04-29
Readership
113
Share



Recently, the Phase II clinical trial results of GS1-144 (GenSci074) were offically published in Menopause, titled Efficacy and safety of GS1-144, a novel neurokinin 3 receptor antagonist, for moderate-to-severe vasomotor symptoms associated with menopause in Chinese women: a randomized, double-blind, placebo-controlled phase 2 trial. The study was led by Peking Union Medical College Hospital(PUMCH), Chinese Academy of Medical Sciences.

This trial was led by Professor Qi Yu from Peking Union Medical College Hospital as the Principal Investigator (PI). It is the first randomized, double-blind, placebo-controlled trial in the Chinese population to confirm the significant efficacy of an NK3R antagonist for moderate-to-severe VMS. The trial met its predefined co-primary endpoints, demonstrated favorable safety and tolerability, and no liver safety risks were observed. The available evidence supports advancing this candidate into confirmatory clinical trials.
With clinical support from PUMCH and GenSci’s innovative R&D, the trial was conducted through close collaboration of 46 clinical centers nationwide. A total of 276 postmenopausal women with ≥7 moderate-to-severe hot flashes per day were enrolled. Eligible subjects were randomized 1:1:1:1 to receive GS1-144 30 mg once daily (QD), 60 mg QD, 30 mg twice daily (BID), or placebo, administered orally for 12 consecutive weeks.
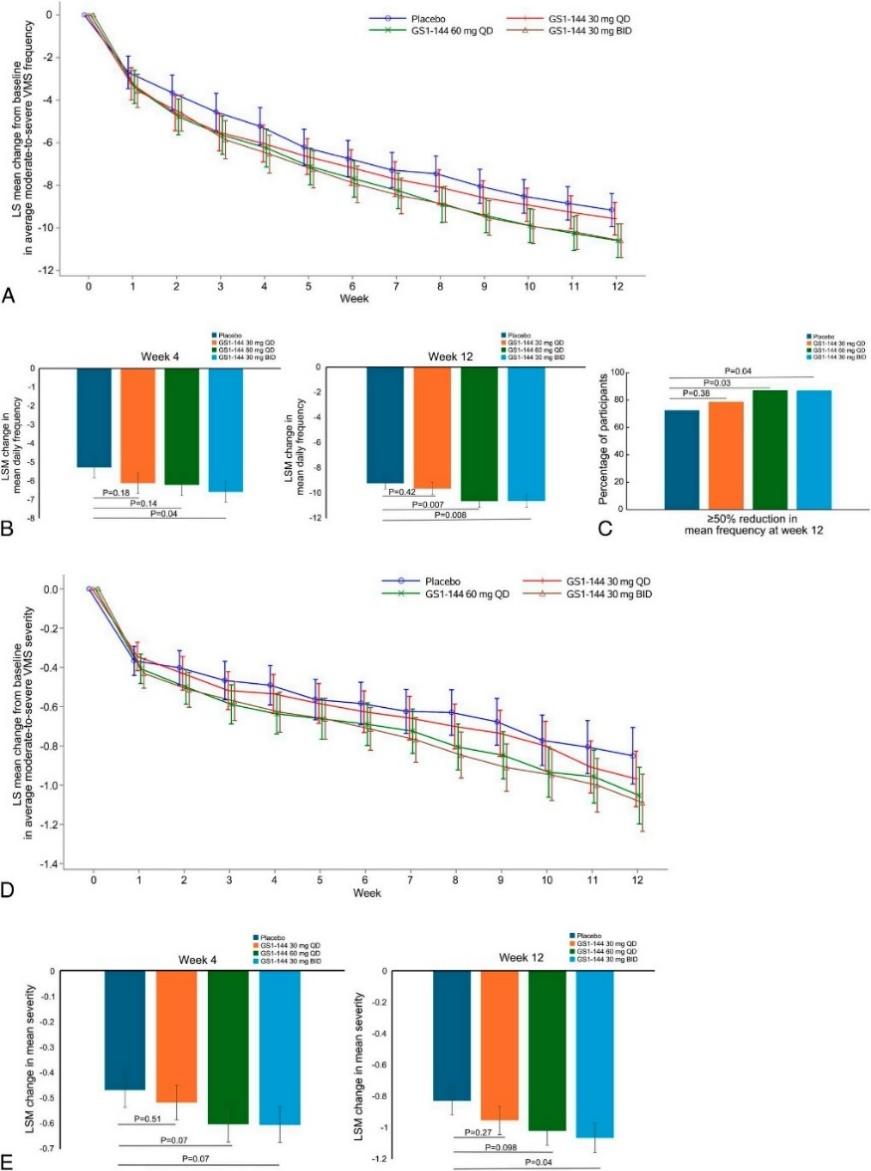
The results showed that, compared with the placebo group:
At Week 4, the mean daily frequency of moderate-to-severe VMS was reduced in the GS1-144 30 mg BID group (least squares mean [LSM] difference −1.305 episodes/day; 90% CI −2.348, −0.263; p = 0.0398), with a clear reduction trend in the 60 mg QD group; At Week 12, both the GS1-144 60 mg QD and 30 mg BID groups showed statistically significant reductions in VMS frequency from baseline (60 mg QD: LSM difference −1.418 episodes/day; 90% CI −2.283, −0.553; p = 0.0072; 30 mg BID: LSM difference −1.407 episodes/day; 90% CI −2.277, −0.536; p = 0.0081); At Week 12, 87% of subjects in the 60 mg QD and 30 mg BID groups achieved a ≥50% reduction in moderate-to-severe VMS frequency from baseline, representing meaningful improvement; For symptom severity, the GS1-144 60 mg QD and 30 mg BID groups also showed significant reductions in mean daily VMS severity at Week 4 and Week 12 versus placebo (30 mg BID: LSM difference −0.237; 90% CI −0.428, −0.046; p = 0.0418; 60 mg QD: LSM difference −0.192; 90% CI −0.382, −0.001; p = 0.0980).
For safety, the incidence of treatment-emergent adverse events (TEAEs) was comparable between the GS1-144 group (67.6%) and the placebo group (62.3%). Most TEAEs were mild or moderate in severity. No definitive liver safety risks were observed during the study period, and no risks of liver enzyme elevation were observed, providing critical safety support for non-hormonal therapy.
Vasomotor symptoms (VMS) are the most common menopausal symptoms, characterized by hot flashes and night sweats. Symptoms persist for 6–10 years in half of women with moderate-to-severe cases, impairing quality of life and mental health([1])([2]). While hormone therapy is effective, some women decline it due to contraindications or safety concerns, and existing non-hormonal treatments show limited efficacy. GS1-144 is poised to offer a breakthrough non-hormonal treatment option for these patients.
Professor Qi Yu, stated: “The findings of GS1-144 are encouraging for Chinese postmenopausal women suffering from moderate-to-severe VMS such as hot flashes and night sweats, and are expected to redefine the Chinese standard for non-hormonal pharmacotherapy. We look forward to GS1-144’s further development to help women navigate menopause safely and comfortably, and restore their quality of life.”
About GS1-144
(GenSci074)GS1-144 is a novel, highly selective NK3R antagonist. It normalizes the activity of overactivated KNDy neurons by blocking overactivated NK3R on KNDy neurons, thereby directly modulating neuronal activity in the thermoregulatory center, reducing abnormal heat signal transmission, and alleviating hot flashes and night sweats. GenSci is actively advancing the subsequent clinical development of GS1-144 to deliver a new non-hormonal treatment option for menopausal women in China and globally.
About GenSci
GenSci is a leading biopharmaceutical company in China, specialising in pediatric and women's health. Additionally, GenSci is active in four other therapeutic areas: Endocrinology, Metabolic, Immunology/Respiratory, and Oncology. The company has over 9,000 employees, and integrates research, development, production, and marketing of innovative therapies in its value chain. Established in 1997, the company is a subsidiary of Changchun High-Tech Industries (Group) and is headquartered in Changchun, China.
Disclosure Notice:
1. Innovative drug research and development entails high risks and uncertainties.
2. This press release is provided as a medical information source for healthcare experts only and is not used for commercial purposes.
3. GenSci does not recommend the use of any unapproved drugs or indications.
References:
[1] Thurston RC. Vasomotor symptoms and cardiovascular health: findings from the SWAN and the MsHeart/MsBrain studies. Climacteric 2024;27:75-80. doi:10.1080/13697137.2023.2196001
[2] Chair SY, Lo SWS, Cheung HY, et al. Vasomotor symptoms, cardiovascular risk factors, and cardiovascular disease risk among Chinese postmenopausal women in Hong Kong. Women Health 2022;62:621-632. doi:10.1080/03630242.2022.2100034