GenSciP166 Selected as Late-Breaking Abstract at SID 2026
Time
2026-05-15
Readership
125
Share



GenSci’s research on GenSciP166, an oral STAT6-targeted protein degrader developed for atopic dermatitis (AD) and other allergic diseases, has been selected as a Late-Breaking Abstract at the 83rd Annual Meeting of the Society for Investigative Dermatology (SID), held in Chicago, USA from May 13 to 16, 2026, and the data will be disclosed for the first time there.
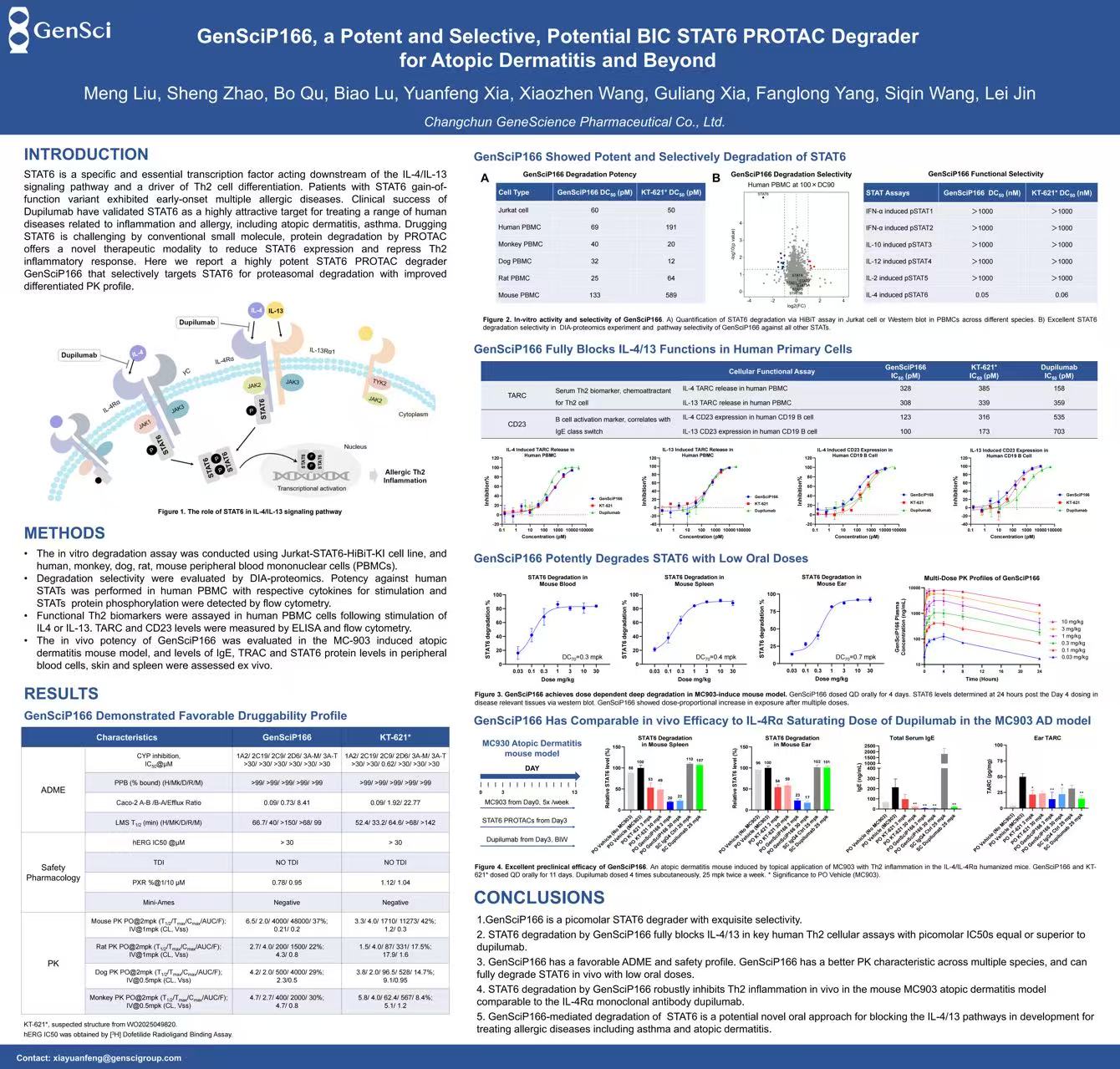
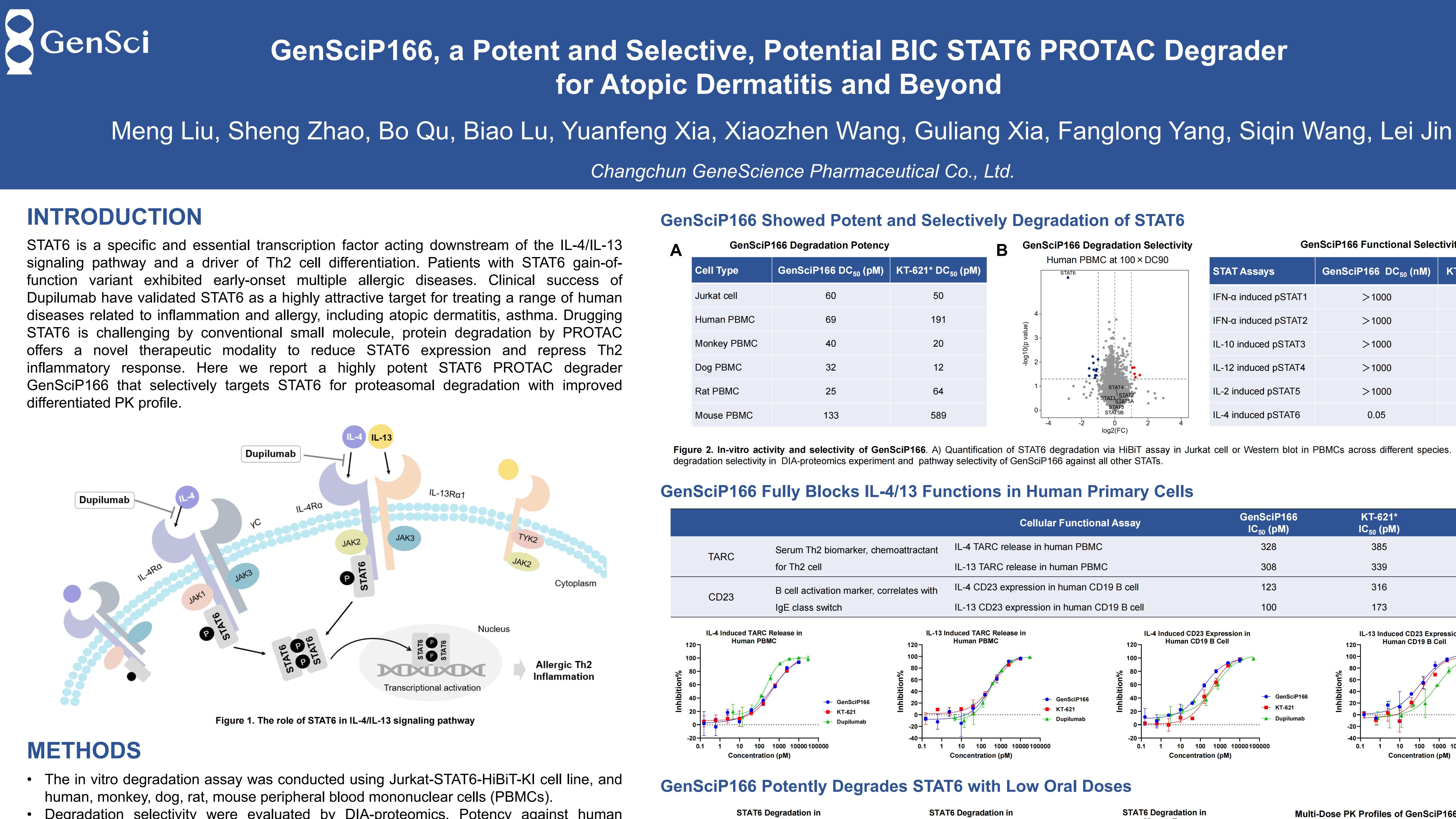
More than 230 million people worldwide living with atopic dermatitis[1], yet existing therapies still face limitations including inadequate response or safety concerns. Signal Transducer and Activator of Transcription 6 (STAT6), an essential transcription factor acting downstream of the IL-4/IL-13 signaling pathway and a central driver of Th2 inflammation in allergic diseases like atopic dermatitis and asthma, has long been considered “undruggable” by conventional small molecules. Proteolysis-targeting chimeras (PROTACs) have opened an completely new avenue to overcome this challenge.
GenSci’s Protein Degrader technology platform integrates multiple technologies including PROTAC, molecule glues, and ATAC to drug “undruggable” intracellular and extracellular proteins. Using the platform’s PROTAC technology, GenSci developed the oral STAT6 degrader GenSciP166. In vitro, GenSciP166 exhibited superior STAT6 degradation and IL-4/IL-13 signaling blockade compared with reference compounds, along with exquisite selectivity for the STAT6 pathway.
In an atopic dermatitis mouse model, low oral doses of GenSciP166 achieved near-complete STAT6 degradation in disease-relevant tissues, matching or exceeding the in vivo suppression of Th2 biomarkers seen with reference compounds. Furthermore, the differentiated pharmacokinetic profile of GenSciP166 suggests its potential to become a best-in-class therapeutic option for atopic dermatitis and beyond.
"Presenting GenSciP166 on a global academic stage like SID is a key opportunity to showcase the potential of our pipeline and the strength of our innovative technology platforms," said Dr. Yuanfeng Xia, Vice President of GenSci and Head of R&D BD License-out. "As we continue to broaden our footprint in immunotherapy and beyond, we welcome global partnerships to bring transformative treatments to patients faster."
Reference: