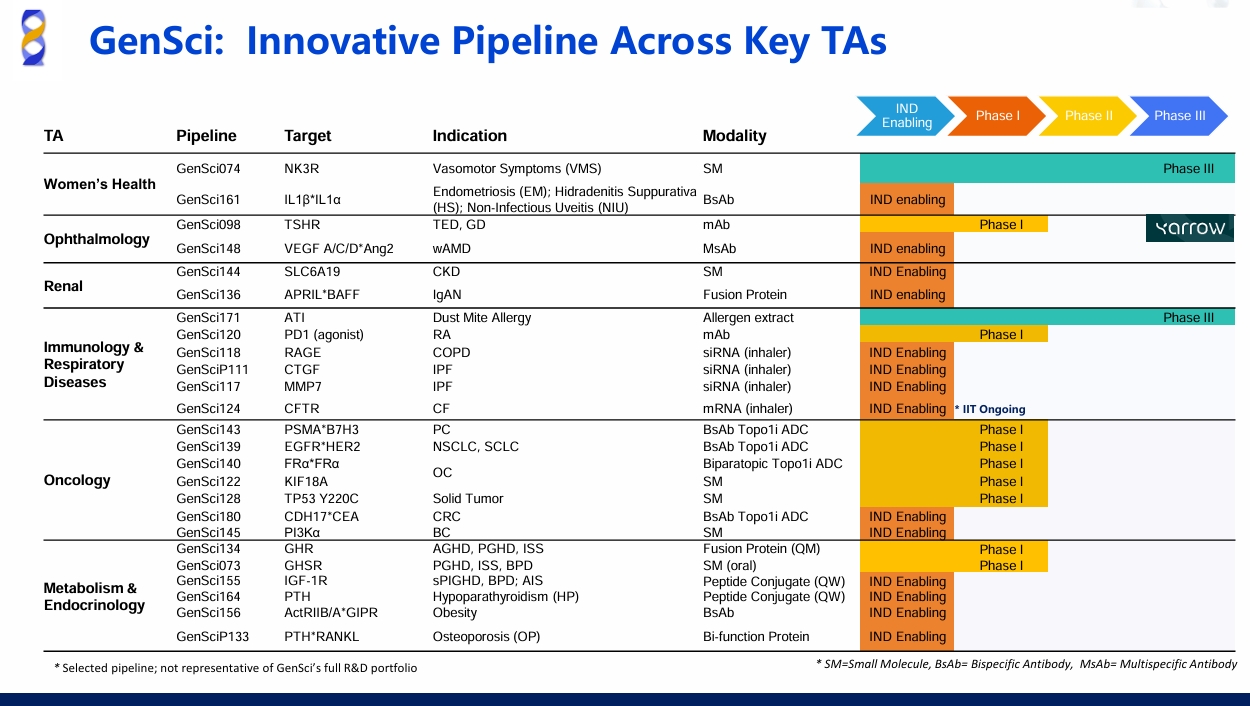
GenSci Presents Multiple Innovative Oncology Pipelines at AACR 2026
Time
2026-04-27
Readership
95
Share



From April 17 to 22, the American Association for Cancer Research Annual Meeting 2026 (AACR 2026), the oldest and largest cancer research organization dedicated to accelerating the conquest of cancer, convened in San Diego, the United States. Changchun GeneScience Pharmaceuticals Co., Ltd. ("GenSci") marked its prominent participation with a series of innovative oncology pipelines under development. Covering cutting-edge technological directions such as bispecific antibody-drug conjugates (BsADC) and multispecific T-cell engagers (TCE), GenSci fully demonstrated its in-depth strategic layout and differentiated innovation strengths in the oncology treatment sector.
Six High‑Potential Oncology Pipelines
GenSci has established technological advantages in the ADC field. Among the six innovative oncology pipelines showcased at the conference, a number of bispecific ADCs (BsADC) leverage HydroLock™, the Company’s proprietary ADC technology platform, to deliver synergistic improvements in precise targeting and safety profiles. In addition, its innovative small molecule therapeutics and trispecific T-cell engagers (TCEs) antibodies exhibit robust differentiated competitive strengths.
GenSci139: EGFR × HER2 BsADC
GenSci139 is a potential first-in-class bispecific antibody-drug conjugate (BsADC) targeting both EGFR and HER2. GenSci139 utilizes a proprietary, highly stable and hydrophilic cleavable linker to conjugate a potent topoisomerase I (TOPO I) inhibitor payload. Combining potent payload activity and excellent plasma stability from its proprietary linker, GenSci139 delivers robust tumor-killing efficacy while presenting a favorable safety profile. In addition, its unique dual-target binding capability markedly enhances tumor cell binding and internalization, while also delivering potent direct cytotoxicity and bystander killing effects.
In multiple mouse CDX models, GenSci139 has showed significantly potent in vivo antitumor efficacy and possesses broad therapeutic potential across various solid tumors, including urothelial carcinoma, lung cancer, gastric cancer, colorectal cancer, ovarian cancer and breast cancer. To support dose selection for the first-in-human (FIH) study (NCT07230977), a multiscale whole-body physiologically based pharmacokinetics (PBPK)-quantitative systems pharmacology (QSP) model was developed to predict the efficacious dose range in patients with advanced solid tumors.
GenSci140: Biparatopic FRα ADC
GenSci140 is a potential best-in-class biparatopic ADC targeting folate receptor alpha (FRα). It adopts a distinctive design incorporating a Fab fragment and a single-domain heavy-chain antibody (VHH) that recognize different FRα epitopes, and utilizes the same proprietary cleavable linker and TOPOI inhibitor payload as GenSci139. Irrespective of FRα expression levels, GenSci140 exhibits significant antitumor efficacy and a favorable potential safety profile in various CDX and PDX models, including ovarian cancer models. Currently, this candidate has been approved for clinical trials in advanced solid tumors by NMPA. A multiscale whole-body physiologically based pharmacokinetics/pharmacodynamics (PBPK/PD) model has been developed to support dose selection.
GenSci180: CDH17 × CEACAM5 BsADC
GenSci180 (formerly known as GS24-B025) is a novel BsADC targeting CDH17 and CEACAM5. Both CDH17 and CEACAM5 are highly expressed tumor-associated antigens in colorectal cancer, and its synergistic dual-targeting design can significantly enhance therapeutic efficacy.
As the third most common cancer worldwide, colorectal cancer has limited treatment options for its metastatic stage (mCRC) . Preclinical studies have demonstrated high co-expression of CDH17 and CEACAM5 in tissue samples from 209 CRC patients. GenSci180 exerts nanomolar affinity and sub-nanomolar cytotoxic activity, delivers superior in vivo anti-tumor efficacy compared with single-target ADCs, and exhibits favorable pharmacokinetic properties. At present, this candidate is steadily advancing IND-enabling studies and is expected to overcome the treatment bottlenecks of advanced colorectal cancer.
GS24-B057: "Best-in-Class" dual-targeting ADC
GS24-B057 is a potential "Best-in-Class" dual-targeting ADC targeting both Nectin‑4 and Trop‑2. Preclinical studies display markedly stronger in vitro cell binding and internalization than single-target ADCs, delivering robust direct and bystander cytotoxic effects in tumor cell lines. In multiple models covering urothelial carcinoma, triple-negative breast cancer and non-small cell lung cancer, GS24-B057 achieves equivalent efficacy to single-target agents combination therapy at lower dose levels and frequency, while maintaining favorable pharmacokinetic properties and a favorable safety profile.
GenSci145: Highly Selective PI3Kα Mutant Inhibitor
GenSci145 is a highly selective PI3Kα mutant inhibitor and represents a second-generation PI3Kα inhibitor, which mechanistically avoids the metabolic side effects of traditional inhibitors. Preclinical studies have verified that this candidate exhibits prominent antitumor activity in a variety of PIK3CA mutant tumor models with excellent blood-brain barrier penetration and effective inhibition of brain metastases. At present, GenSci145 is conducting Phase1 clinical trials in China, and the IND submission to FDA is planned for the first half of 2026.
GS24-B047: "Best-in-Class" DLL3-targeted trispecific TCE
GS24-B047 is a highly promising Best-in-Class DLL3-targeted TCE. Serving as a molecular bridge between tumor cells and immune cells, it binds DLL3 on small cell lung cancer (SCLC) tumor cells, CD3 on T cells, and CD2 as a co-stimulatory receptor to form a stable immunological synapse. Preclinical studies have demonstrated that GS24-B047 exhibits enhanced in vitro T cell dependent cellular cytotoxicity, accompanied by reduced cytokine release and a favorable safety profile. It delivers superior antitumor efficacy in SCLC CDX models and prolongs overall survival. Studies in mice and non-human primates have both demonstrated favorable pharmacokinetic characteristics and acceptable safety.
As the world’s most influential academic event in the field of oncology, the 2026 AACR Annual Meeting gathers top-tier global research breakthroughs. More than 100 Chinese pharmaceutical enterprises showcased nearly 400 research achievements at the conference, fully highlighting the global competitiveness of China’s innovative drug industry.Fu Li, Senior Director of Cancer Biology at GenSci, emphasized, “GenSci will deepen strategic cooperation with world’s leading research institutions and pharmaceutical companies, accelerate the global clinical development and commercialization of our R&D pipeline, and bring more Chinese innovative medicines to benefit patients worldwide.”
Disclosure Notice
1. Innovative drug research and development entails high risks and uncertainties.
2. This press release is provided as a medical information source for healthcare experts only and is not used for commercial purposes.
3. GenSci does not recommend the use of any unapproved drugs or indications.