GenSci128, an Investigational Cancer Therapy from Gensci Pharmaceuticals, Receives FDA Orphan Drug Designation for Pancreatic Cancer
Time
2026-03-11
Readership
110
Share



Shanghai, China – March 11, 2026 – Changchun GeneScience Pharmaceutical Co., Ltd. ("GenSci") recently announced that the U.S. Food and Drug Administration (FDA) has granted Orphan Drug Designation (ODD) to its novel investigational therapeutic, GenSci128, for the treatment of pancreatic cancer. This marks a significant regulatory milestone for the GenSci128 program, accelerating its global clinical advancement. It also underscores Gensci's commitment to aligning its innovative R&D capabilities with international standards.
Orphan Drug Designation is a special status conferred by the FDA to facilitate the development of therapies for rare diseases affecting fewer than 200,000 individuals in the United States. With this designation, GenSci128 is now eligible for various development and commercial incentives, including more frequent interactions and protocol assistance with the FDA, partial tax credits for clinical trial costs, waiver of applicable user fees, and a potential seven-year period of market exclusivity in the U.S. upon approval.
Pancreatic cancer remains one of the most aggressive and lethal malignancies, characterized by high invasiveness and a dismal prognosis, with a five-year survival rate of under 10% globally. The Orphan Drug Designation for GenSci128 not only has the potential to expedite its clinical development and regulatory review in the U.S. but also aims to mitigate development costs and risks, potentially paving the way for a much-needed breakthrough therapeutic option for patients battling the disease.
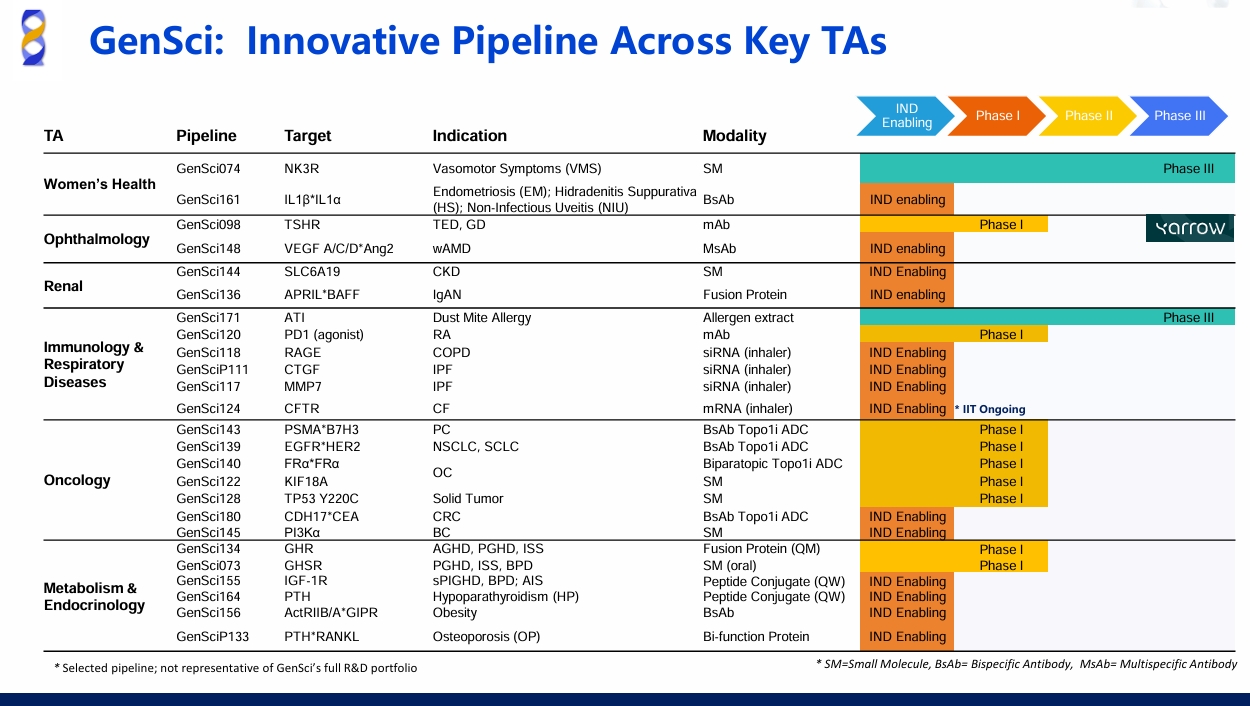
GenSci128 is a small molecule designed as a selective reactivator of the p53 protein specifically targeting the TP53 Y220C mutation. The TP53 gene, encoding the crucial tumor suppressor protein p53, is the most frequently mutated gene in human cancers. While the TP53 Y220C mutation constitutes approximately 1.8% of all TP53 mutations, there are currently no approved targeted therapies addressing this specific mutation globally, representing a significant area of high unmet medical need. GenSci128 is designed to bind selectively to a pocket created by the Y220C mutation, stabilizing the p53 protein in its wild-type conformation and restoring its normal transcriptional activity and tumor-suppressing functions. Preclinical studies have demonstrated promising efficacy and a favorable safety profile for GenSci128.
Classified as a new chemical entity under the U.S. 505(b)(1) regulatory pathway, GenSci128 is being developed for the treatment of locally advanced or metastatic solid tumors harboring the TP53 Y220C mutation, including pancreatic, ovarian, breast, and colorectal cancers. The company has already received clearance for its Investigational New Drug (IND) application from the FDA, as well as approval from Chinese authorities to initiate clinical trials for these indications.
The FDA's Orphan Drug Designation for GenSci128 is a key achievement reflecting Gensci's strategic commitment to building a globally competitive, innovative pipeline. The company has established a robust portfolio of cutting-edge technology platforms, including Antibody-Drug Conjugates (ADCs), small molecule targeted therapies, siRNA, and long-acting release formulations. Within the oncology arena, Gensci is actively developing a diversified portfolio of clinical-stage assets. This includes GenSci139, a potential first-in-class EGFR/HER2 bispecific ADC; GenSci140, a dual-epitope ADC targeting FRα; and GenSci145, a novel mutant-selective PI3Kα inhibitor. These programs collectively demonstrate Gensci's focused depth and strategic breadth in addressing critical challenges in solid tumor treatment.
-
 2026-03-11GenSci128, an Investigational Cancer Therapy from Gensci Pharmaceuticals, Receives FDA Orphan Drug Designation for Pancreatic Cancer
2026-03-11GenSci128, an Investigational Cancer Therapy from Gensci Pharmaceuticals, Receives FDA Orphan Drug Designation for Pancreatic Cancer -

 2026-01-16GenSci Unveils 7 Innovative Pipeline at JPM 2026
2026-01-16GenSci Unveils 7 Innovative Pipeline at JPM 2026 -

 2025-12-16GenSci Secures Up to $1.365B in Global ex-China Licensing Deal for GS-098(YB-101) with RTW Investments
2025-12-16GenSci Secures Up to $1.365B in Global ex-China Licensing Deal for GS-098(YB-101) with RTW Investments





